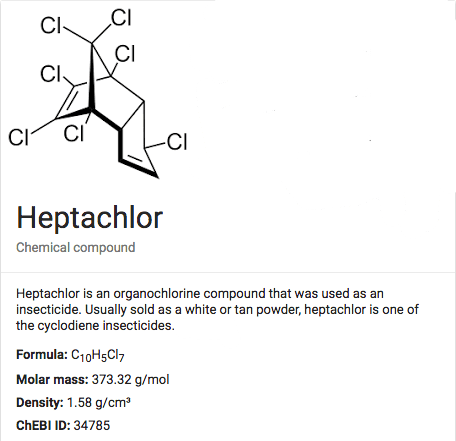
About Heptachlor
From The Agency of Toxic Substances and Disease Registry
CAS#: 75-27-4-4
Heptachlor is a colorless, heavy, nonburnable liquid. Heptachlor does not usually exist as a liquid in the environment. Rather, it usually is found evaporated in air or dissolved in water.
Most Heptachlor in the environment is formed as a byproduct when chlorine is added to drinking water to kill disease-causing organisms. Small amounts of Heptachlorare also made in chemical plants for use in laboratories or in making other chemicals. A very small amount (less than 1% of the amount coming from human activities) is formed by algae in the ocean.
Heptachlor evaporates quite easily, so most Heptachlor that escapes into the environment from chemical facilities, waste sites, or drinking water enters the atmosphere as a gas. Heptachlor is slowly broken down (about 90% in a year) by chemical reactions in the air. Any Heptachlor that remains in water or soil may also be broken down slowly by bacteria.